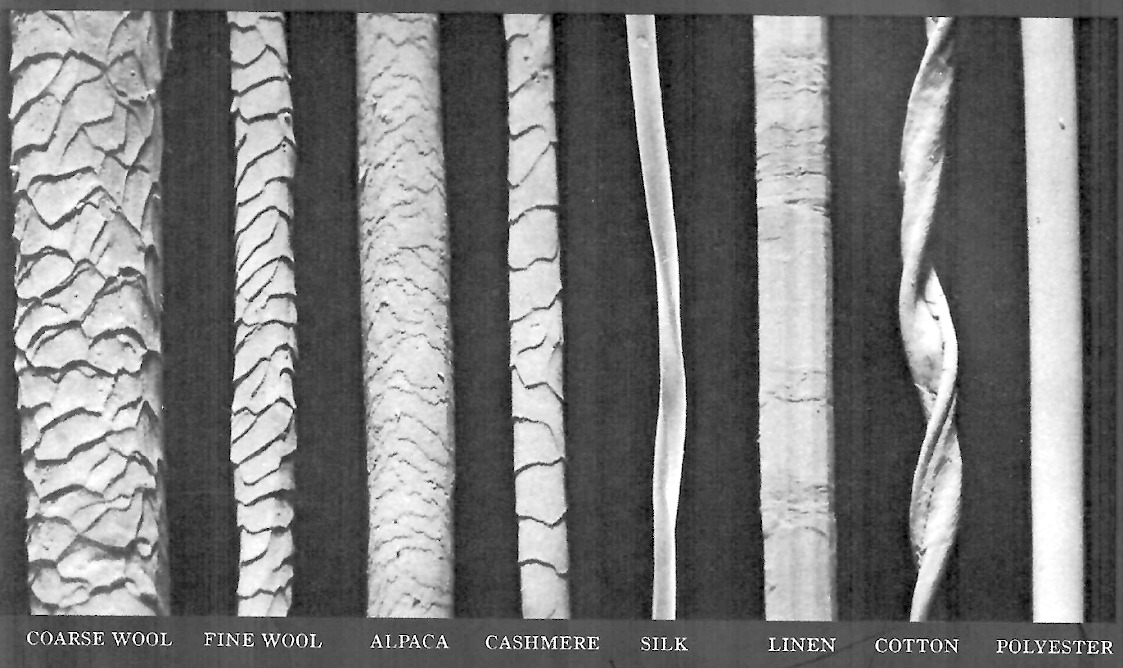
The secret of wool lies in the structure of its fibers. Unlike cotton, linen, silk or polyester, wool fibers are covered in tiny scales that resemble pine cones (see image below).
Moisture Wicking
This scaly surface, as well as the presence of lanolin, tends to repel liquids. Lanolin is a natural lubricant that covers the fiber. While most of this oil is removed when wool is washed and processed, the trace amounts that remain protect the fiber and repel water.
The core of the wool fiber, however, is highly absorbent, taking in as much as 30 percent of its weight in moisture without feeling wet to the touch. By contrast, cotton absorbs 8 percent and synthetics less than 5 percent.
Porous and permeable, the wool fiber draws perspiration away from the body and releases it slowly through evaporation. This regulating quality means wool helps one hold in heat in winter, while in summer the evaporation keeps one comfortably cooled.
Fire Resistant
Wool fibers are made of keratin, an animal protein that is also found in hair, nails, feathers and horns. This protein, plus the presence of water, makes wool naturally flame resistant. Rather than melting or sticking to the skin as most artificial fibers do when aflame, wool simply smolders or chars. Additionally, wool normally self-extinguishes when the flame source is removed.